Nelson Labs - What considerations need to be taken when creating a Biological Evaluation Plan? This #TBT video from Dr Sophie Michel - Associate Biocompatibility Expert, looks at the crucial first step

تويتر \ ICON Plc على تويتر: "The ISO 10993-1 standard means that manufacturers need a structured plan for the biological safety evaluation of their #medicaldevice. Explore how ICON's team of experts can

Biological Evaluation Plan: A crucial first step in the Biocompatibility evaluation of a Med Device - YouTube
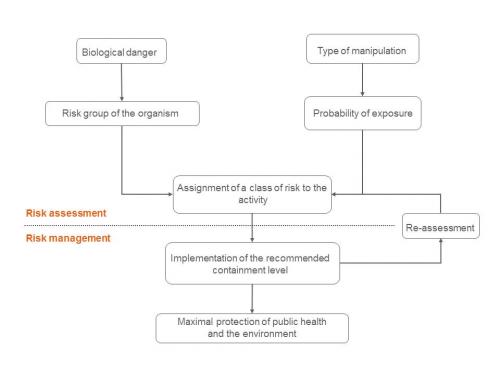
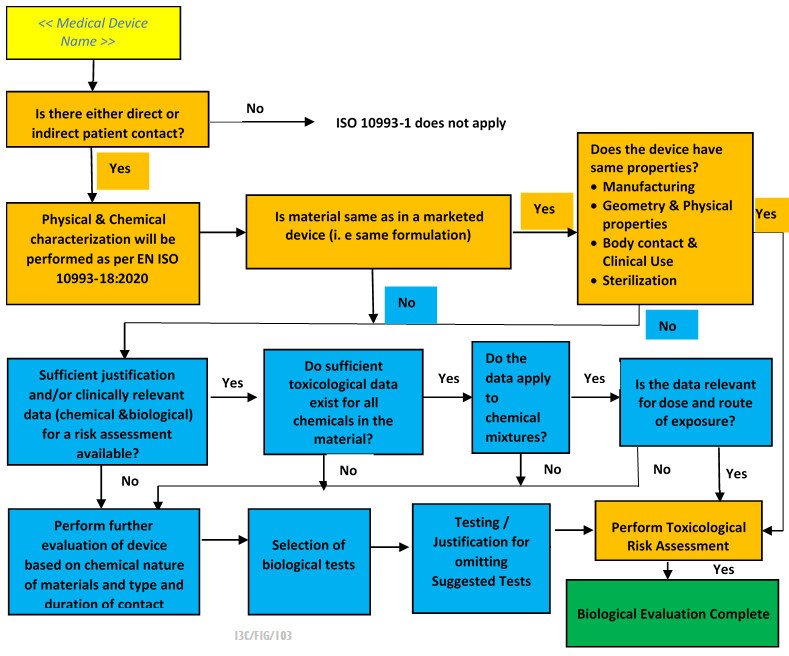
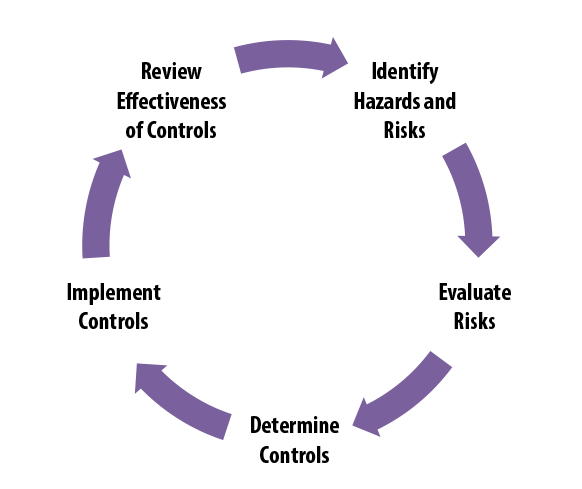
Suggested Contents for Biological Evaluations and Biological Assessments When you prepare a Biological Evaluation (BE) or Biolog