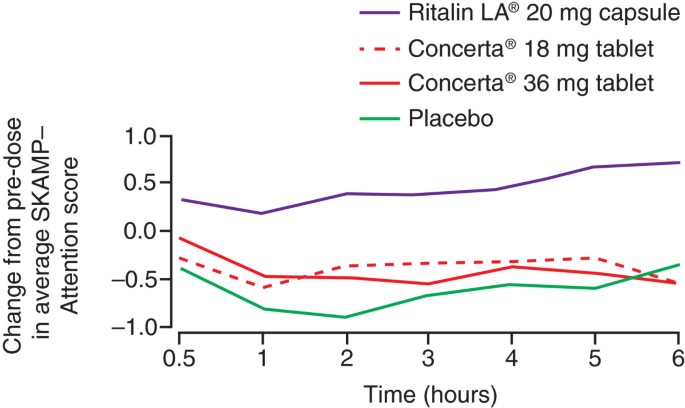
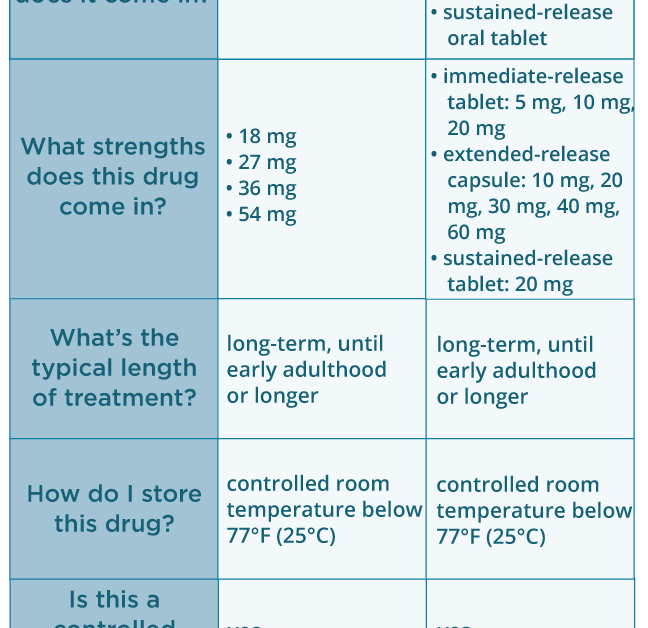
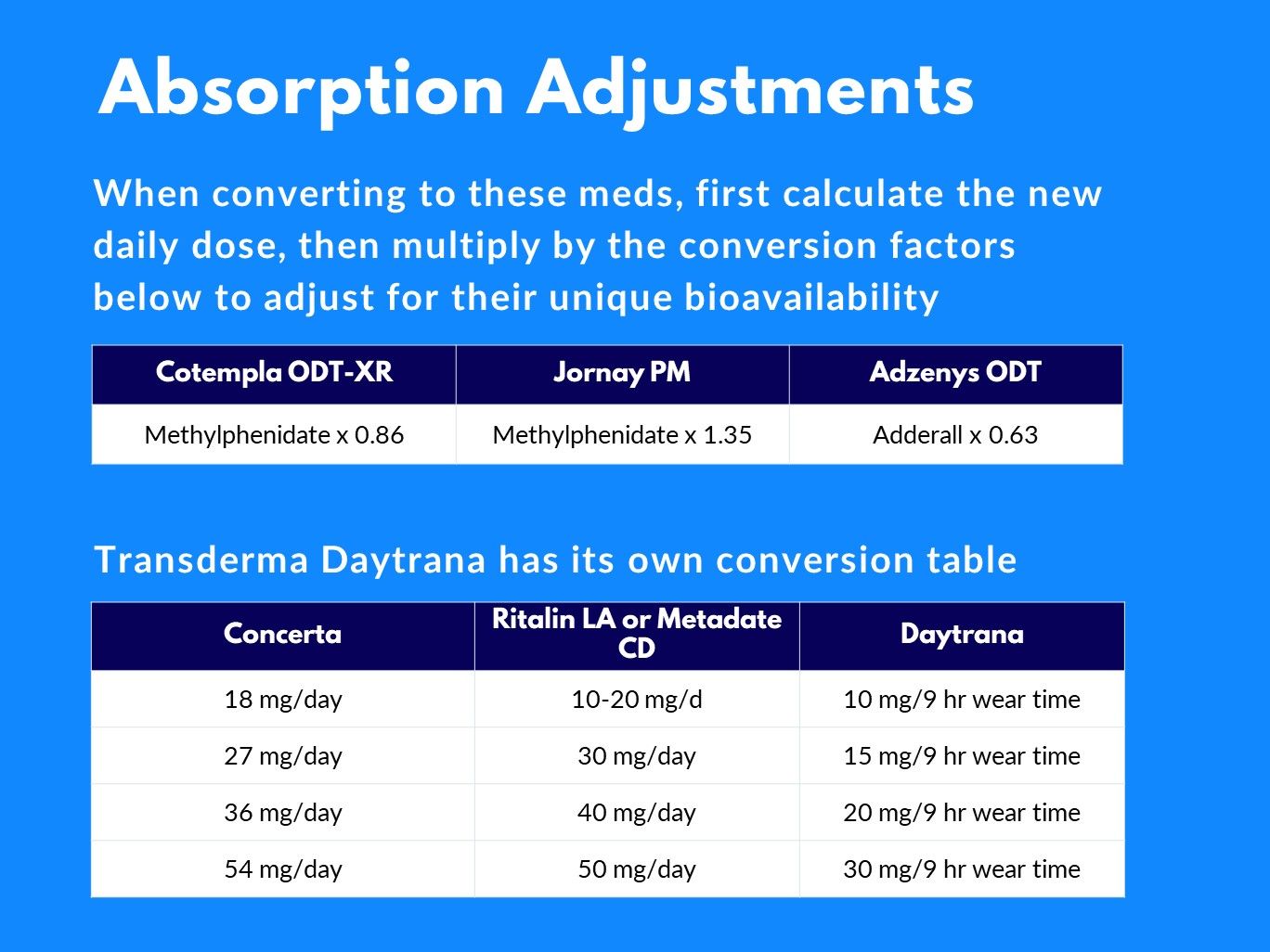
A Post Hoc Comparison of Prior ADHD Medication Dose and Optimized Delayed-release and Extended-release Methylphenidate Dose in a Pivotal Phase III Trial - Clinical Therapeutics

WO2018085256A1 - Effective dosing of a child for the treatment of adhd with methylphenidate - Google Patents