Safety and tolerability of AAV8 delivery of a broadly neutralizing antibody in adults living with HIV: a phase 1, dose-escalation trial | Nature Medicine

Low-dose mRNA-1273 COVID-19 vaccine generates durable memory enhanced by cross-reactive T cells | Science
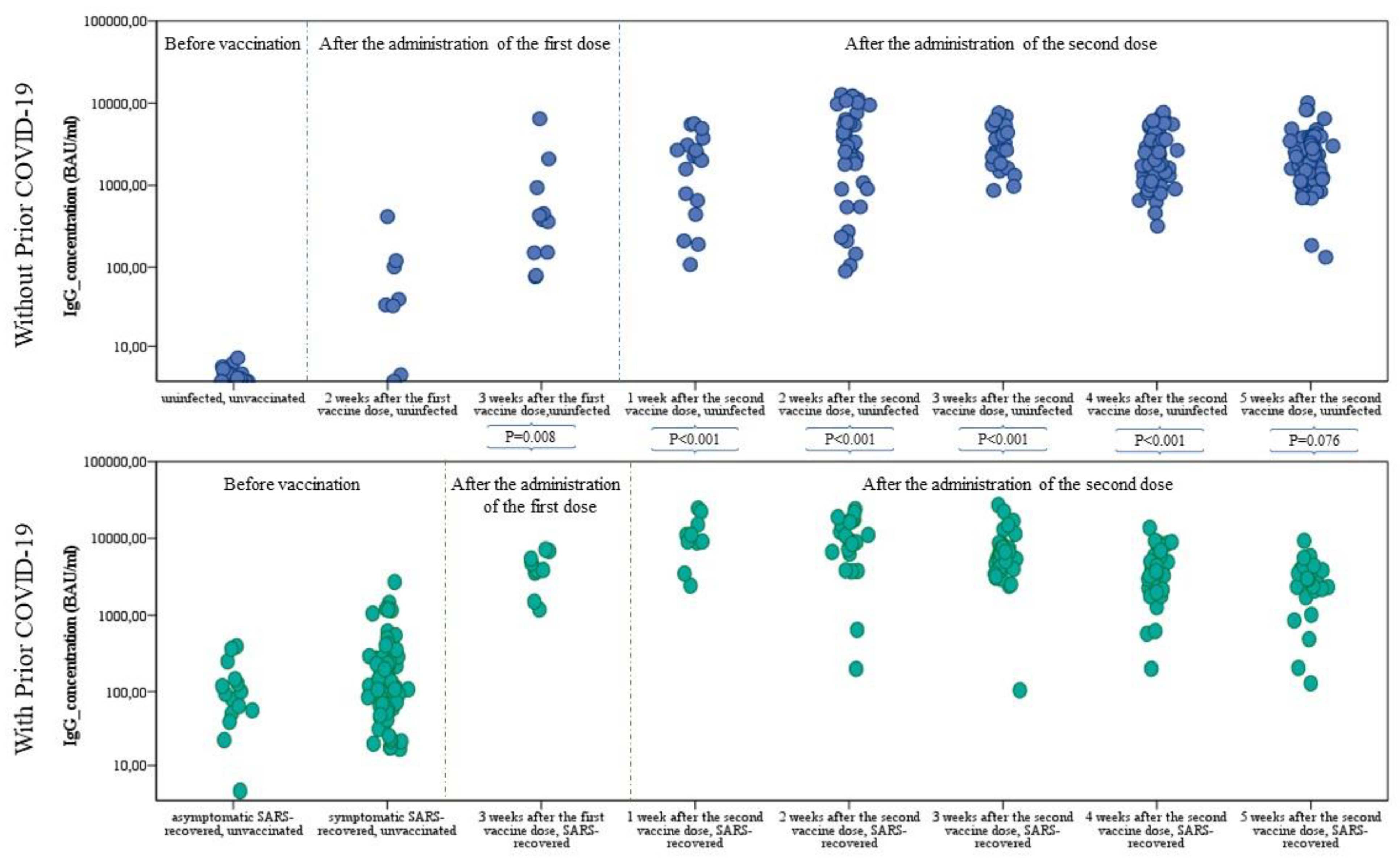
Cells | Free Full-Text | Differences in the Concentration of Anti-SARS-CoV-2 IgG Antibodies Post-COVID-19 Recovery or Post-Vaccination | HTML
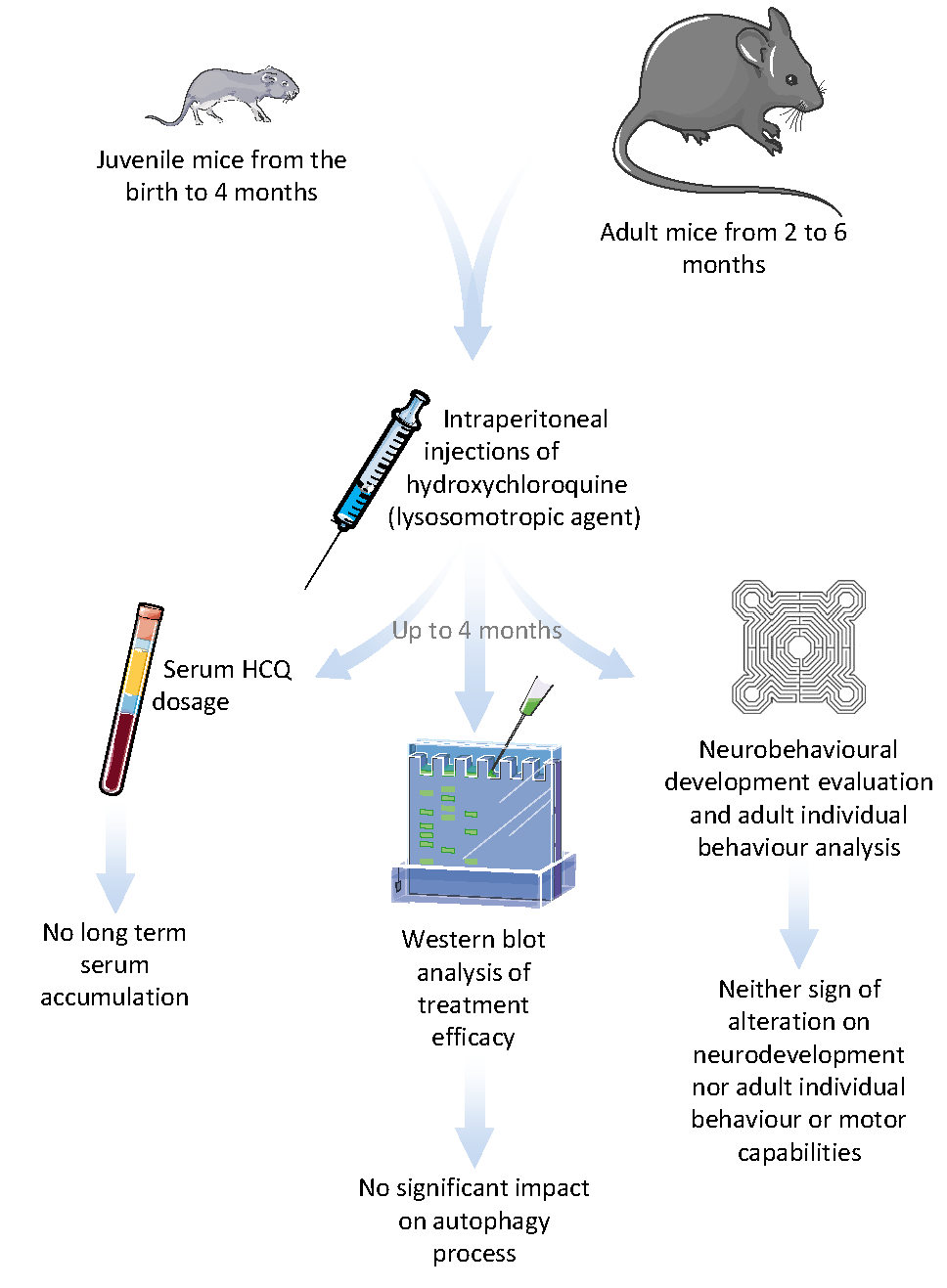
Biomedicines | Free Full-Text | Long Term Pharmacological Perturbation of Autophagy in Mice: Are HCQ Injections a Relevant Choice? | HTML
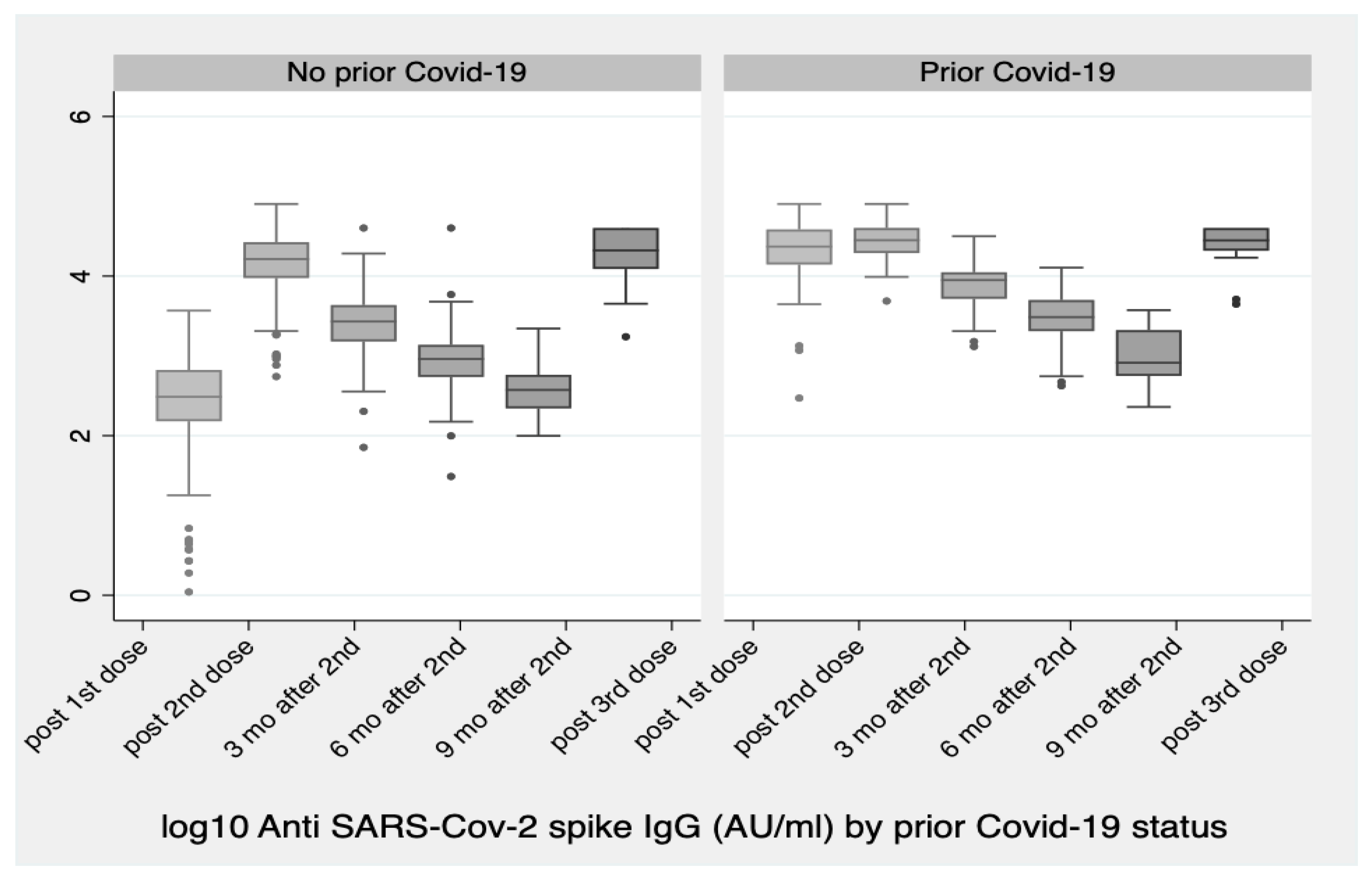
Vaccines | Free Full-Text | Significant Increase in Antibody Titers after the 3rd Booster Dose of the Pfizer–BioNTech mRNA COVID-19 Vaccine in Healthcare Workers in Greece | HTML

Two doses of the SARS-CoV-2 BNT162b2 vaccine enhance antibody responses to variants in individuals with prior SARS-CoV-2 infection | Science Translational Medicine

Safety and tolerability of AAV8 delivery of a broadly neutralizing antibody in adults living with HIV: a phase 1, dose-escalation trial | Nature Medicine

An AAV-based, room-temperature-stable, single-dose COVID-19 vaccine provides durable immunogenicity and protection in non-human primates - ScienceDirect

TropicalMed | Free Full-Text | Humoral Immune Response Induced by the BBIBP-CorV Vaccine (Sinopharm) in Healthcare Workers: A Cohort Study | HTML

Robust immune responses are observed after one dose of BNT162b2 mRNA vaccine dose in SARS-CoV-2–experienced individuals | Science Translational Medicine

mRNA vaccination boosts cross-variant neutralizing antibodies elicited by SARS-CoV-2 infection | Science

Safety and immunogenicity of seven COVID-19 vaccines as a third dose (booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV-BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial -
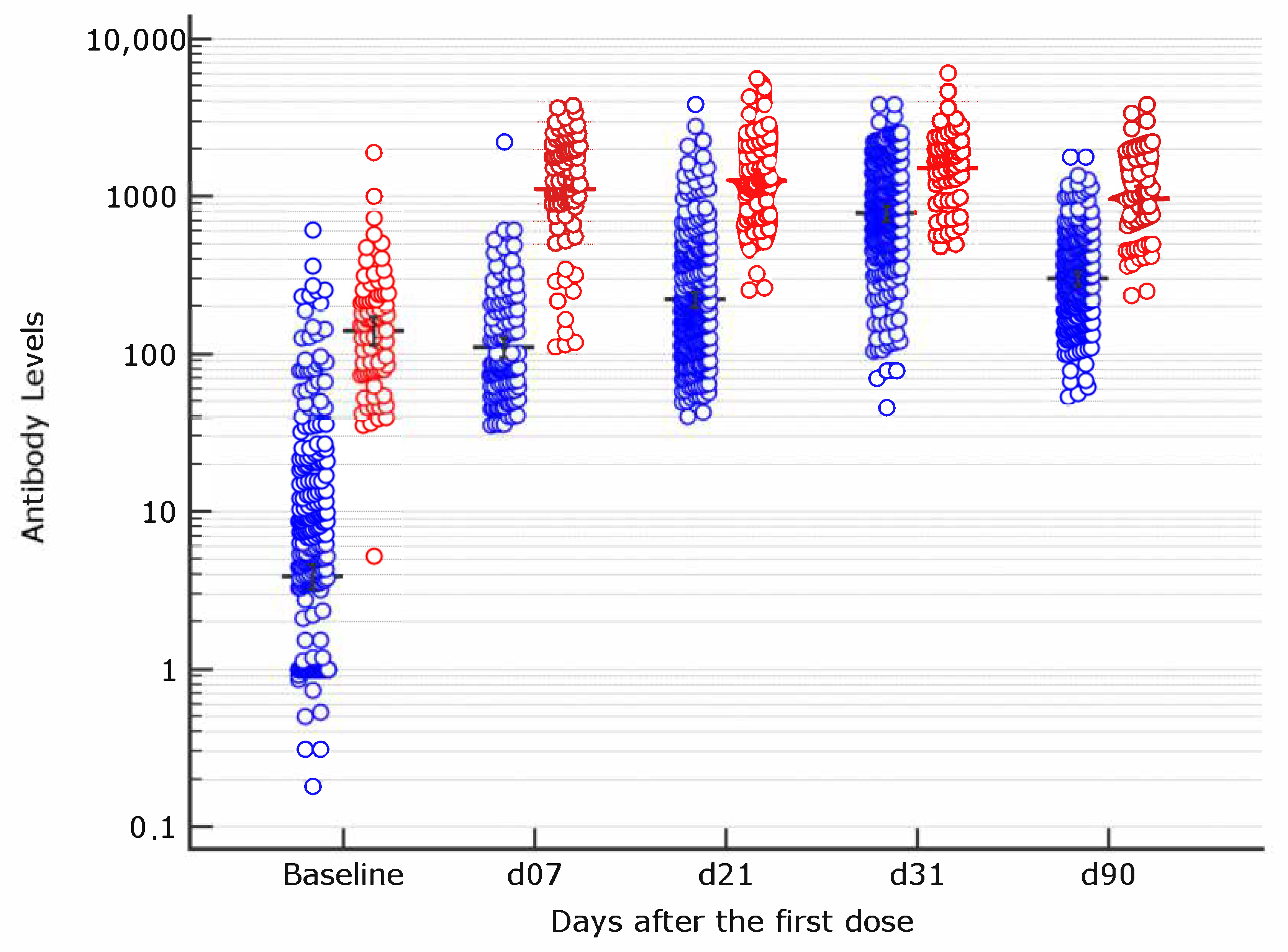
Vaccines | Free Full-Text | Early Serological Response to BNT162b2 mRNA Vaccine in Healthcare Workers | HTML

Tranexamic acid for prevention of hemorrhage in elective repeat cesarean delivery—a randomized study - American Journal of Obstetrics & Gynecology MFM

Low-Dose Sorafenib Acts as a Mitochondrial Uncoupler and Ameliorates Nonalcoholic Steatohepatitis - ScienceDirect

Immunogenicity of mRNA-1273 COVID vaccine after 6 months surveillance in health care workers; a third dose is necessary - Journal of Infection
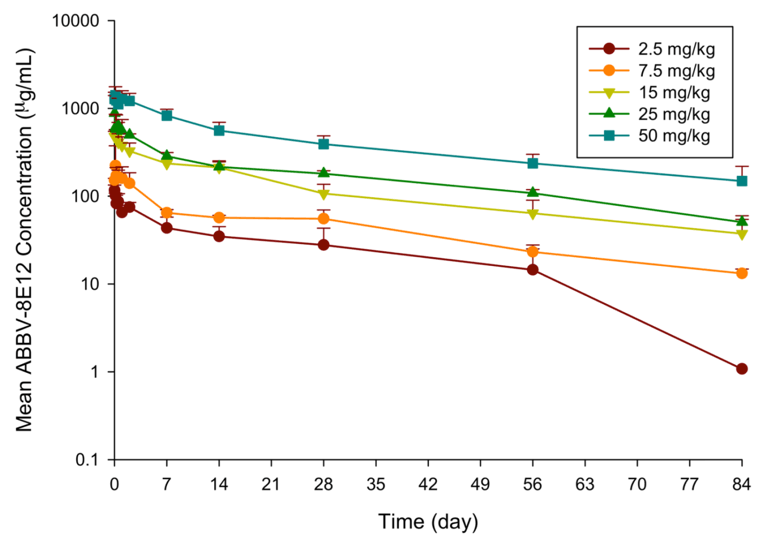
PRECLINICAL AND CLINICAL DEVELOPMENT OF ABBV-8E12, A HUMANIZED ANTI-TAU ANTIBODY, FOR TREATMENT OF ALZHEIMER'S DISEASE AND OTHER TAUOPATHIES • The Journal of Prevention of Alzheimer's Disease

Safety and immunogenicity of seven COVID-19 vaccines as a third dose (booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV-BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial -

Robust immune responses are observed after one dose of BNT162b2 mRNA vaccine dose in SARS-CoV-2–experienced individuals | Science Translational Medicine
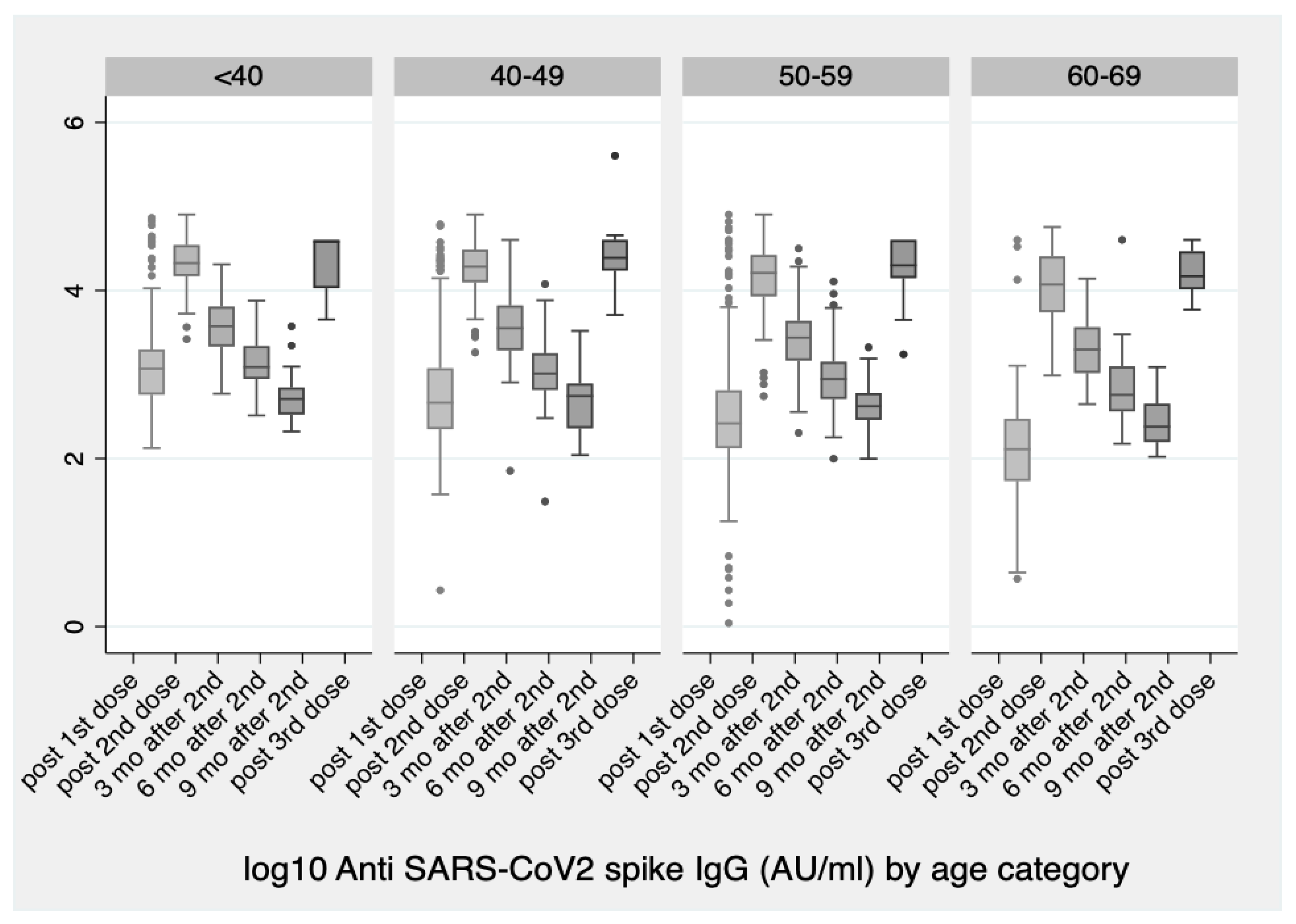
Vaccines | Free Full-Text | Significant Increase in Antibody Titers after the 3rd Booster Dose of the Pfizer–BioNTech mRNA COVID-19 Vaccine in Healthcare Workers in Greece | HTML

Omecamtiv mecarbil does not prolong QTc intervals at therapeutic concentrations - Trivedi - - British Journal of Clinical Pharmacology - Wiley Online Library

A Single-Dose Intranasal ChAd Vaccine Protects Upper and Lower Respiratory Tracts against SARS-CoV-2 - ScienceDirect