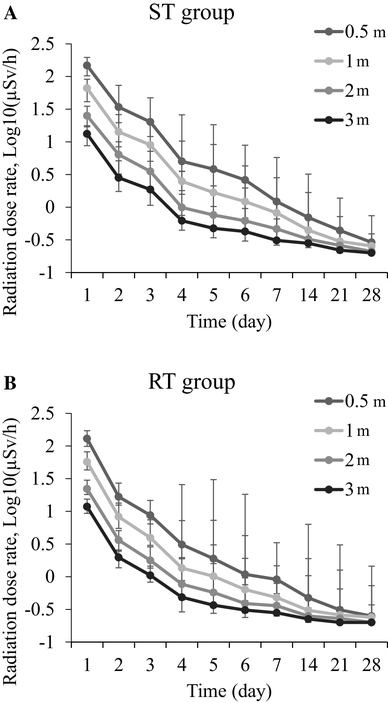
Assessment of potentially reusable edible wild plant and mushroom gathering sites in eastern Fukushima based on external radiation dose - ScienceDirect

Predicting late radiation-induced xerostomia with parotid gland PET biomarkers and dose metrics - Radiotherapy and Oncology

G Live on Twitter: "Vaccinations Update: 18-39 yr olds can walk-in & get the Pfizer vaccine @GLiveGuildford until 7pm today. Pfizer also available for 18-39 yr olds on Fri 8am-7pm & Sat

Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK - The Lancet

Hindustan Times al Twitter: "The DCGI approved Biological E's coronavirus vaccine #Corbevax as a heterologous #COVID19 booster dose (also known as precaution shot) for all adults. Read more here: https://t.co/1incjIfkJm https://t.co/OU0e4xGUnK" /

Safety, immunogenicity, and reactogenicity of BNT162b2 and mRNA-1273 COVID-19 vaccines given as fourth-dose boosters following two doses of ChAdOx1 nCoV-19 or BNT162b2 and a third dose of BNT162b2 (COV-BOOST): a multicentre, blinded,
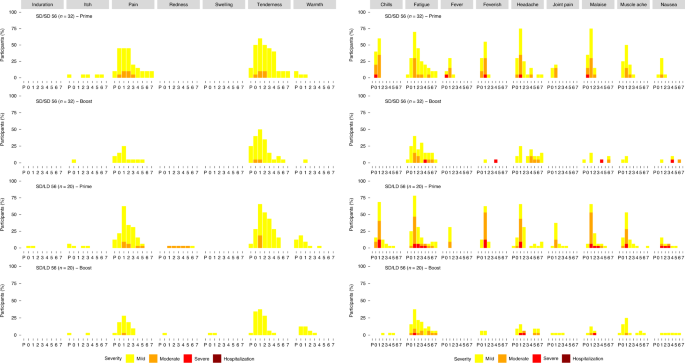
Phase 1/2 trial of SARS-CoV-2 vaccine ChAdOx1 nCoV-19 with a booster dose induces multifunctional antibody responses | Nature Medicine
Deep Learning–based Reconstruction for Lower-Dose Pediatric CT: Technical Principles, Image Characteristics, and Clinical Impl

Safety and immunogenicity of seven COVID-19 vaccines as a third dose (booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV-BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial -
Dose-related effect of trisodium nitrilotriacetate monohydrate on renal tumorigenesis initiated with N-ethyl-N-hydroxyethyl- nit

Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials - The Lancet

Safety and immunogenicity of the ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 in people living with and without HIV in South Africa: an interim analysis of a randomised, double-blind, placebo-controlled, phase 1B/2A trial -

Reactogenicity and immunogenicity after a late second dose or a third dose of ChAdOx1 nCoV-19 in the UK: a substudy of two randomised controlled trials (COV001 and COV002) - The Lancet
Deep Learning–based Reconstruction for Lower-Dose Pediatric CT: Technical Principles, Image Characteristics, and Clinical Impl
Highly pathogenic avian influenza virus H5N1 controls type I IFN induction in chicken macrophage HD-11 cells: a polygenic trait that involves NS1 and the polymerase complex | Virology Journal | Full Text

Persistence of immunogenicity after seven COVID-19 vaccines given as third dose boosters following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK: Three month analyses of the COV-BOOST trial. - Journal